Products
Ursofalk
Ursofalk (ursodeoxycholic acid, UDCA) is used for the treatment of Primary Biliary Cholangitis (PBC), for the dissolution of radiolucent gallstones and for hepatobiliary disorders associated with Cystic Fibrosis. UDCA helps stimulate impaired hepatobiliary secretion, protects the biliary epithelium against toxic effects of hydrophobic bile acids and protects hepatocytes from apoptosis.
Ursofalk is available in both solid and liquid oral formulations:
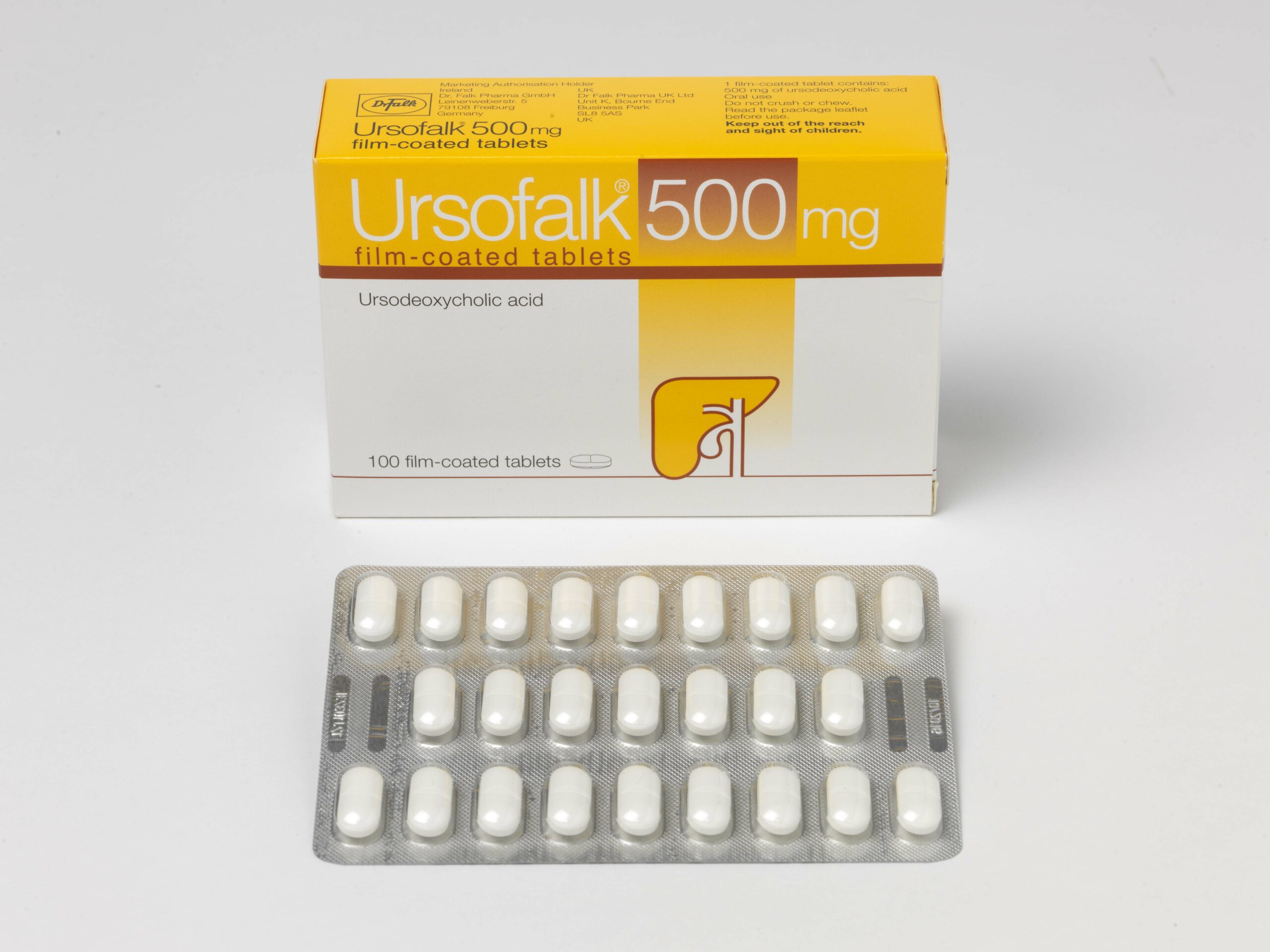
- Ursofalk 500mg tablets
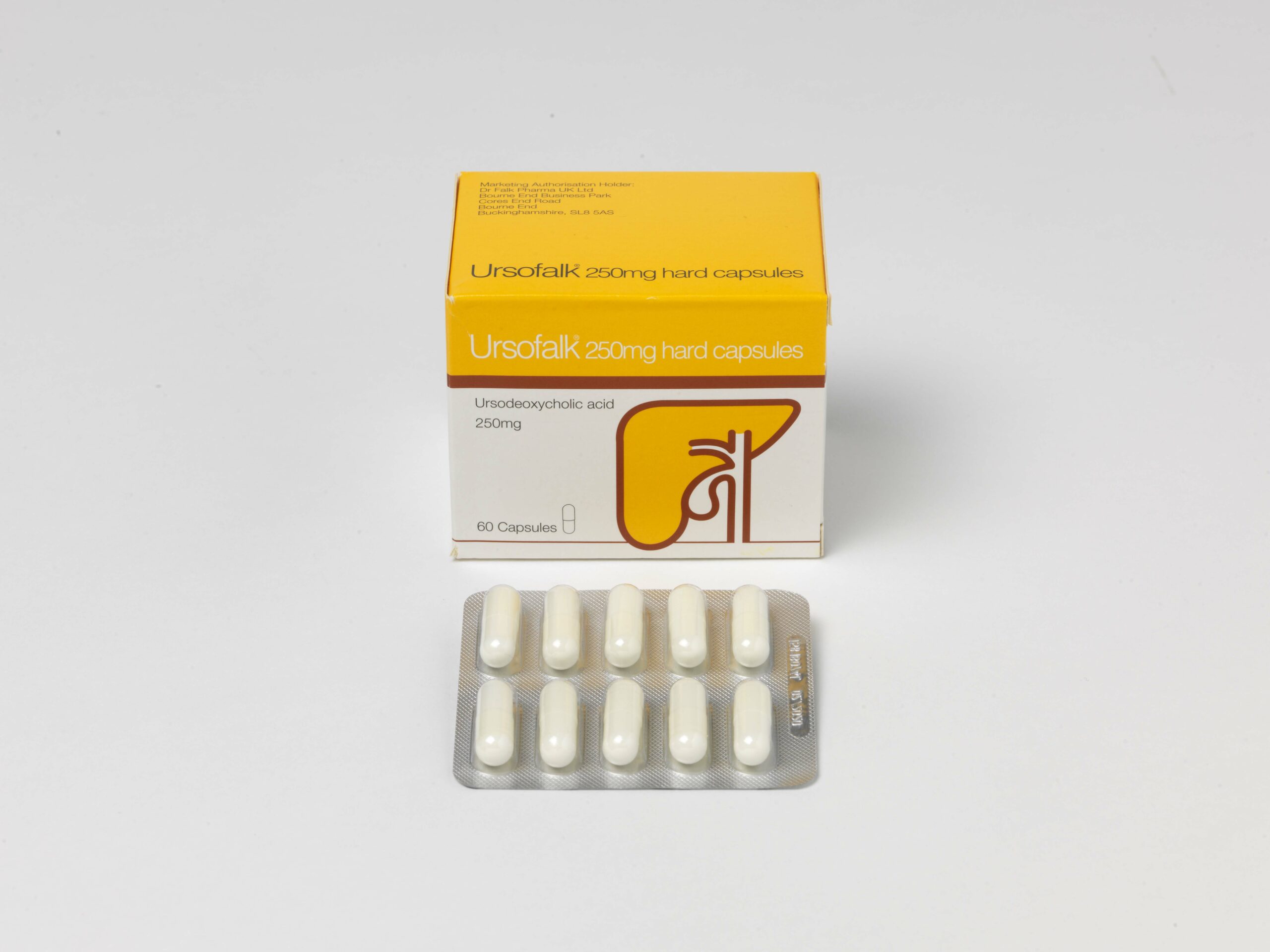
- Ursofalk 250mg capsules
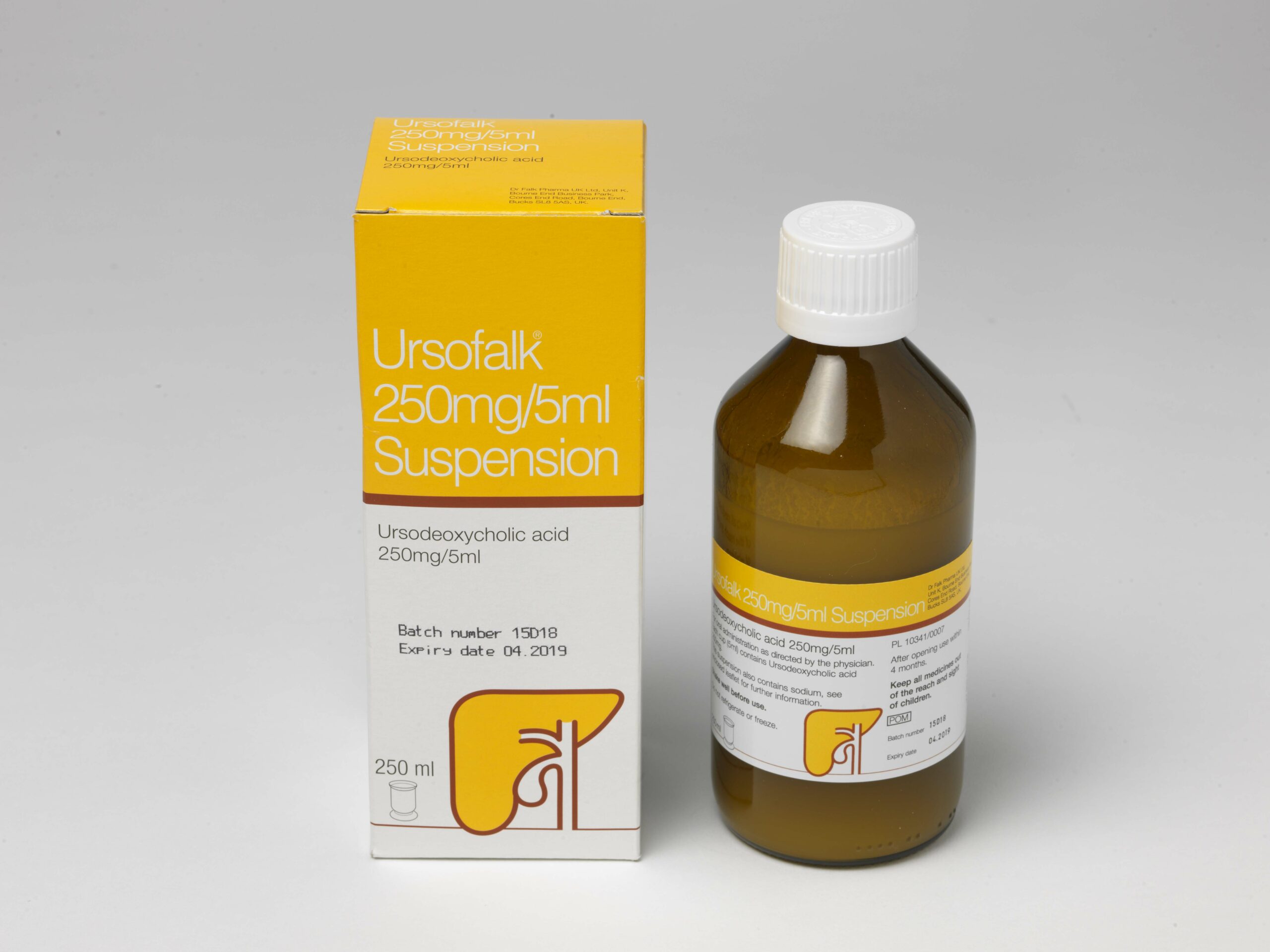
- Ursofalk 250mg lemon flavoured suspension formulation to make swallowing easier.
Discover more about Ursofalk
These convenient and flexible Ursofalk formulations are designed specifically to improve patient adherence in this lifelong condition.
Prescribing Information (Refer to full SPC before prescribing).
Presentations: Ursofalk 250mg hard capsules containing 250mg ursodeoxycholic acid (UDCA), Ursofalk 250mg/5ml suspension, containing 250mg UDCA per 5ml suspension, Ursofalk 500mg film-coated tablets containing 500mg UDCA. Indications: Treatment of primary biliary cirrhosis (cholangitis) (PBC) where there is no decompensated hepatic cirrhosis; stages I-III. Dissolution of radiolucent cholesterol gallstones of less than 15mm, in patients with a functioning gall bladder. Paediatric population: hepatobiliary disorders associated with cystic fibrosis in children aged 6 to 18 years (capsules and tablets) or 1 month to 18 years (suspension). Dosage: For PBC – adults and elderly: 12-16mg UDCA/kg/day in 3 divided doses during the first three months and once daily in the evening when liver values improve. For gallstones: approximately 10mg UDCA/kg/day taken in the evening. Follow up cholecystograms may be useful. Children: PBC and gallstones are rare in children and adolescents. Data are limited. Dose should be related to body weight and condition. For hepatobiliary disorders associated with cystic fibrosis in children aged 1 month to 18 years: 20 mg/kg/day in 2-3 divided doses, with a further increase to 30 mg/kg/day if necessary. Contra-indications: Acute inflammation of the gall bladder or biliary tract. Occlusion of the biliary tract. Radio opaque gall bladder. Frequent episodes of biliary colic. Radio-opaque calcified gallstones. Impaired contractility of the gall bladder. Hypersensitivity to bile acids or any excipient of the formulation. When used for hepatobiliary disorders associated with cystic fibrosis in children aged 1 month to 18 years – unsuccessful portoenterostomy or without recovery of good bile flow in children with biliary atresia. Warnings/Precautions: Carry out liver function tests every 4 weeks in the first 3 months of treatment, then every 3 months. Decompensation of hepatic cirrhosis may occur when used for advanced PBC, which partially regresses on discontinuation of treatment. Rarely, in PBC, clinical symptoms may worsen. Reduce dose to 250mg daily and gradually increase to recommended dose. Monitor gallstone dissolution with oral cholecystography, ultrasound control, at 6 – 10 months after starting treatment. Stop treatment in cases of persistent diarrhoea. 5ml suspension contains 11 mg sodium, equivalent to 0.6 % of the WHO recommended maximum daily intake of 2 g sodium for an adult. 5ml suspension also contains 7.5 mg benzoic acid which may increase jaundice in neonates and 50 mg propylene glycol which may induce serious adverse effects in neonates. Interactions: Cholestyramine, colestipol and certain antacids (e.g. aluminium hydroxide, aluminium oxide) bind bile acids and may interfere with absorption/efficacy. Leave a 2-hour gap between taking these substances and Ursofalk. The absorption of ciclosporin may be affected and absorption of ciprofloxacin and nitrendipine reduced. Concomitant UDCA and rosuvastatin resulted in slightly elevated plasma levels of rosuvastatin. Therapeutic effect of dapsone may also be affected. Oestrogenic hormones and cholesterol lowering agents e.g. clofibrate, increase biliary lithiasis. Use in pregnancy: There are no adequate data. Use only in pregnancy if clearly necessary. When treating women of childbearing potential non-hormonal or low oestrogen oral contraceptive measures are recommended. If used for gallstone dissolution, then effective non-hormonal contraception should be used. No adverse reactions are expected in breastfed infants. Undesirable effects: Pasty stools, diarrhea (common). Upper abdominal pain (in PBC treatment), calcification of gallstones, decompensation of hepatic cirrhosis, urticaria (very rarely). Legal category: POM. Cost: Ursofalk 250mg capsules, 100-capsule pack: £31.88; €23.56. 60-capsule pack: £30.17. Ursofalk 250mg/5ml suspension, 250ml bottle: £26.98, €26.66. Ursofalk 500mg tablet, 100-tablet pack: £80; €50.95. Product licence holder: In the UK: Dr Falk Pharma UK Ltd; Unit K, Bourne End Business Park, Cores End Road, Bourne End, SL8 5AS. In Ireland: Dr. Falk Pharma GmbH, Leinenweberstrasse 5, 79108 Freiburg, Germany. Product licence number: Ursofalk capsules: PL10341/0006; PA573/5/1. Ursofalk suspension: PL10341/0007; PA573/5/2. Ursofalk tablets: PL10341/0010; PA573/5/3. Date of preparation: February 2024.
Further information is available on request.
Adverse events should be reported. In the UK, visit www.mhra.gov.uk/yellowcard. In Ireland, visit https://www.hpra.ie/homepage/about-us/report-an-issue/human-adverse-reaction-form Adverse events should also be reported to Dr Falk Pharma UK Ltd at PV@drfalkpharma.co.uk
Summary of Product Characteristics (SmPC)
UK
For further information, the Summary of Product Characteristics (SmPC) are available below.
Republic of Ireland
For further information, the Summary of Product Characteristics (SmPC) are available below.
Patient Resources
This information on the following pages is intended only for UK and Irish residents who have been prescribed either Budenofalk, Salofalk, Ursofalk or Jorveza.

